Our Mission
To enhance our customer's ability to realize operational efficiencies, control costs and reduce risks associated with rapidly evolving regulatory environments by offering flexible, scalable and industry expert quality assurance service solutions.

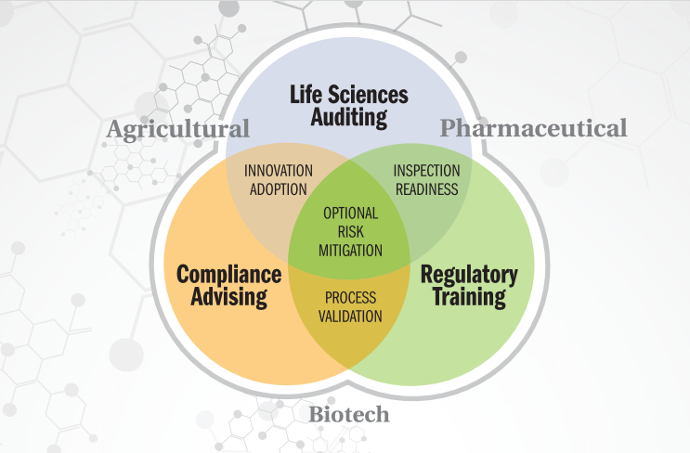
What we do
QA Compliant provides GxP services to the Biotechnology, Pharmaceutical, Animal Health, Agrochemical industries as well as support contract research organizations.
-
Good Laboratory Practice (GLP) - QA Compliant provides your company with comprehensive GLP services including: Standard Operating Procedure development specifically for your laboratory under 21 CFR Part 58 to assure regulatory compliance.
-
Good Clinical Practice (GCP) - QA Compliant provides a full menu of GCP compliance services including SOP reviews, Clinical trial protocol and amendment management, deviation procedures, Trial Master File (TMF) organization and management.
-
Good Manufacturing Practice (GMP) - Our services ensure that the minimum requirements for the production, control and distribution of your products are met. These include auditing facility design, validation, and maintenance, personnel training, documentation, quality control, equipment validation and maintenance, product testing, product release, analytical method validation, process validation and transfer.
Core Values

Integrity
The moral and ethical principles of honesty and trust guide us in all we do.

Value
The work we do enables our clients to flexibly, efficiently, and economically achieve compliance.

Teamwork
Working together in a united way is how we achieve success for our clients and team members.

Wisdom
Our deep-seated knowledge and experience inform every aspect of our business and ensure that we provide the highest-quality regulatory guidance to our clients.

Consistency
The results and solutions we provide are consistently reliable and measurably accurate.

Social Responsibility
Serving the greater good and making a positive impact on our clients, their customers, and our community is of utmost importance to us.